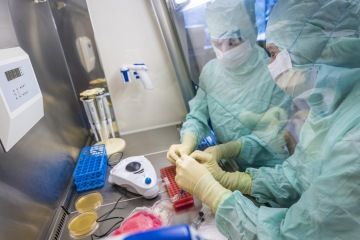
Manufacturing of high quality cell and gene therapy products
SCTbio offers its partners the highest quality services by delivering an engaging, methodical, and cost-effective approach to GMP manufacturing, quality control, process development and logistics services for your cell and gene therapy and viral vector manufacturing needs.
SCTbio Brochure for Analytical Services
Check-out SCTbio full range of analytical capabilities!
SCTbio Brochure
SCTbio Your Reliable Partner for Cell & Gene Therapy GMP Manufacturing
ISCT 2025
May 29 – April 1, 2024 in Vancouver
Cell & Gene on the Med
April 15 - 17, 2025, Roma
German Biotech Days
April 9 - April 10, 2025, Heidelberg Congress Center, Berlin
Advanced Therapies Congress, London
March 18-19, 2025 at ExCel, London
SCTbio and Cyto-care Collaborate to Improve Cell Therapy Cryopreservation and Logistics Capabilities in Europe
Source: Press Release - Prague, Czech Republic and Vienna, Austria
Orgenesis Announces Collaboration Agreement with SCTbio for the Production of Lentivirus Vectors
Source: Press Release - Germantown, MD and Prague, Czech Republic
SCTbio Video
Take a tour of our GMP manufacturing facility in Prague.
Ukázka článku - Media
Praesent nec arcu lorem. Nulla vitae malesuada nisl, nec laoreet enim. Lorem ipsum dolor sit amet, consectetur adipiscing elit.
Cambridge
SOTIO, LLC
200 Cambridgepark Drive, Suite 3100,
Cambridge, MA 02140
USA
Prague
SCTbio a.s.
Jankovcova 2,
170 00 Prague 7
Czech Republic
Bejing
SCTbio a.s.
Jankovcova 2,
170 00 Prague 7
Czech Republic
Manufacturing of high quality cell and gene therapy products
SCTbio offers its partners the highest quality services by delivering an engaging, methodical, and cost-effective approach to GMP manufacturing, quality control, process development and logistics services for your cell and gene therapy and viral vector manufacturing needs.

Manufacturing of high quality cell and gene therapy products
SCTbio offers its partners the highest quality services by delivering an engaging, methodical, and cost-effective approach to GMP manufacturing, quality control, process development and logistics services for your cell and gene therapy and viral vector manufacturing needs.
Leading engine
for the field of cell
and gene therapy
SCTbio is a stand-alone contract manufacturing organization offering full GMP services covering production of Advanced Therapy Medicinal Products (ATMPs) including genetically modified and viral vectors. It is a part of PPF Biotech network. The SCTbio mission is to serve as the leading engine for the rapidly advancing field of cell and gene therapy by offering a superior approach to GMP manufacturing for its partners.
Services
SCTbio offers its capacities in the GMP manufacturing facility in Prague. The SCTbio expert team is available to manage technology transfers of products in any stage of preclinical/clinical development and to accommodate it to high cGMP standards.
cGMP Manufacturing
& Quality Control
Analytical
Development
Process
Development
Logistics Services
& Apheresis Collection
Procurement
& Warehouse Management
Quality & Regulatory Support
& Qualified Person Services
Facilities
SCTbio is proud to be a global organization, operating facilities in two continents which include GMP cell therapy manufacturing facilities in Prague, Czech Republic and Beijing, China. SCTbio has also access to a viral vector manufacturing facility operated by its affiliated company based in Cambridge, MA, USA.
Cambridge
SOTIO, LLC
200 Cambridgepark Drive, Suite 3100,
Cambridge, MA 02140
USA
Prague
SCTbio a.s.
Jankovcova 2,
170 00 Prague 7
Czech Republic
Bejing
Manufacturing Facility SCTbio
Beijing Economic-Technological Dev. Area,
99 Kechuang 14, 30-B,
Beijing 100176, China